Aortic Aneurysm
- What is an abdominal aortic aneurysm?
- Are aortic aneurysms common?
- Why do aortic aneurysms develop?
- Why are aortic aneurysms important?
- Can aortic aneurysms be prevented?
- How will I know if have an aortic aneurysm?
- When do aortic aneurysms require treatment?
- Thoracoabdominal aneurysms
- What treatments are available for aortic aneurysms?
- What are the results of surgery for aortic aneurysms?
- Are there complications of treatment?
- What measures are taken to reduce complications?
- Aortic aneurysm screening programmes
- Aortic aneurysm links
- References
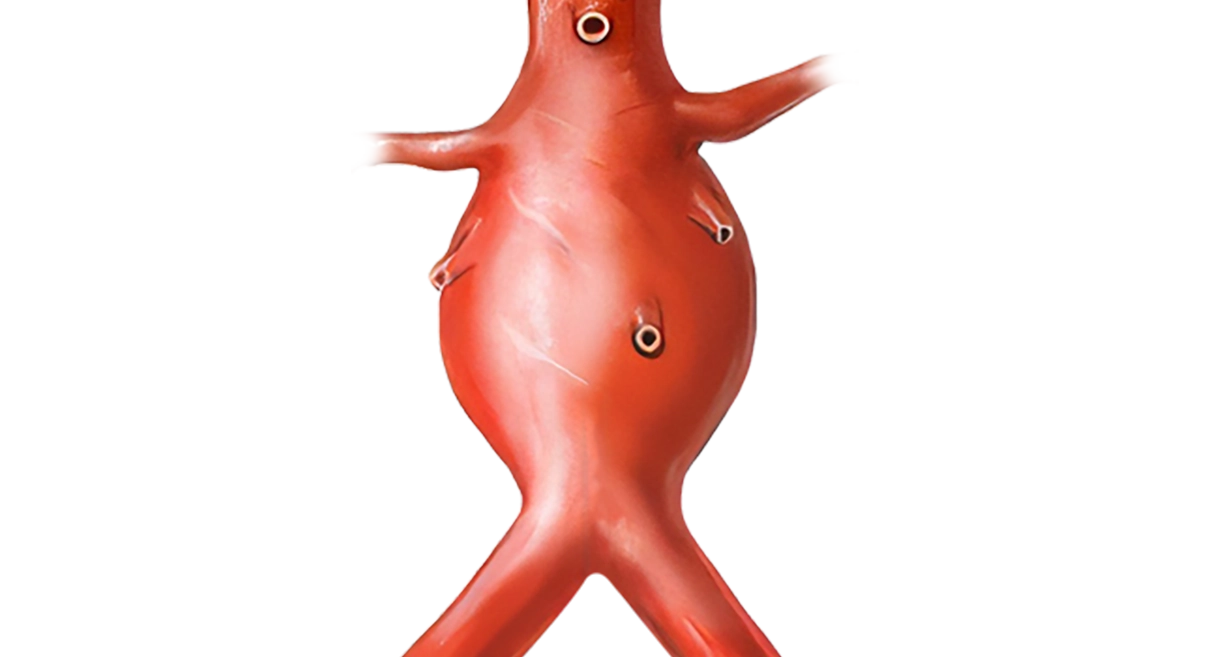
What is an abdominal aortic aneurysm?
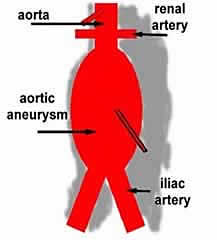
An aneurysm is a swelling or dilatation in a blood vessel. Aneurysms can occur in any blood vessel, but are much more common in arteries, although they do occur rarely in veins. An abdominal aortic aneurysm (AAA) is a dilatation in the abdominal (tummy) part of a major artery – the aorta. This is one of the commonest types of aneurysm.
Are aortic aneurysms common?
Aortic aneurysms are fairly common especially in older men. Hospital admission rates for aneurysms were increasing (Filipovic M et al, 2005) but recent studies indicate that the incidence of aneurysms may now be decreasing (Sandiford et al, 2011). About 6% of men (6 in 100) aged 80 years will have an aneurysm (Bengtsson H et al, 1992). They account for 1.4% of deaths in men over the age of 65 years and 0.5% of deaths in women. About10,000 people die in the UK from a ruptured aortic aneurysm each year (Sayers RD, 2002). They are more common in brothers and there is an increased risk of an aortic aneurysm if you suffer from high blood pressure or atherosclerosis (hardening of the arteries) especially in smokers. Aortic aneurysms seem to be relatively rare in Asian populations. The Maori population in New Zealand has a higher death rate from aneurysms (8.9 per 100,000 in Maori vs 3.7 per 100,000 in non-Maori) and require more emergency operations than the non-Maori population, despite a slightly lower age-adjusted admission rate (12 per 100,000 vs 15 per 100,000, Rosaak JI et al, 2003).
Why do aortic aneurysms develop?
We do not know exactly why some people develop aortic aneurysms. They are much more common in men and may sometimes run in the family. There seems to be an approximately four times increased risk of having an aneurysm for the brother of a patient with an aneurysm. Stated another way the brother of a patient with an aneurysm has about a 10-15% chance of developing an aneurysm. Most brothers (more than 80%) will not develop an aneurysm. Surprisingly, the presence of diabetes seems to have a slight protective effect on aneurysm development. Aneurysms may develop because of a weakness in the tissues holding the blood vessels together or possibly an imbalance in various enzymes (matrix metallo-proteinases or MMPs) that are found in the blood vessel wall. No specific genes have yet been identified in relation to aneurysms (Powell JT, 2003). Inflammation has a role in the development of aneurysms and the latest work has indicated an important role for the protein Cyclophilin A which may trigger an inflammatory response (Wintraub 2009).
Why are aortic aneurysms important?
Aortic aneurysms are important because sometimes they can burst. When an aortic aneurysm bursts it is a catastrophic event in which the patient can die from internal bleeding in a matter of minutes. In most people a burst (ruptured) aortic aneurysm is fatal. The risk of an aneurysm rupturing varies with the aneurysm size. The larger the aortic aneurysm the more risk of it rupturing. Small aneurysms less than 5.5cms in diameter have an annual risk of rupture of less than 1% (1 in 100). This means that a patient, with an aortic aneurysm less than 5.5cms in diameter, has approximately a 1 in 100 chance of it bursting over the next 12 months. A recent Canadian study has reported specific figures for the risk of rupture based on aneurysm size (Brown PM et al 2003). For men the annual risk of an aneurysm rupturing was 1-1.8% for aneurysms between 5.0 and 5.9cms, but increased to 14.1-15.6% when the aneurysm was 6cms or greater. In other words a man with a 5.4cms diameter aneurysm has only 1 or 2 chances in a hundred that his aneurysm will rupture in the next year. Once the aneurysm increases to 6.1cms that risk will increase to approximately 15 chances in a hundred. In women the risks are greater. A woman with an aneurysm between 5.0 and 5.9 cms has a 3.9 – 4.7% risk of rupture over the next year. Once the aneurysm size increases beyond 6.0 cms the risk of rupture increases to 22 – 30% over the next year.
For individuals driving for domestic use: an individual with a thoracic aneurysm of greater than 6cm diameter or an abdominal aneurysm of greater than 5.5cm, or another vascular abnormality at risk of dissection or rupture, is generally considered unfit to drive. Individuals with Marfans syndrome should not drive if they have an aneurysm of greater than 4.5cm
NZ Transport Agency regulations
Less commonly aortic aneurysms can cause other symptoms. The aneurysm is usually lined by blood clot, which in most people, is not dangerous. Occasionally, parts of this blood clot can be dislodged and travel downwards to block arteries to the leg (embolism). As the aneurysm enlarges it can cause pressure on nerves and can occasionally lead to pressure on the ureter (the tube between the kidney and bladder). This can prevent urine draining from the kidneys normally and the kidneys can become damaged. Repair of an aortic aneurysm will stop these complications developing.
Can aortic aneurysms be prevented?
At present aortic aneurysms cannot be prevented from developing, but their growth may be slowed by some simple measures. If you smoke, this increases the rate of growth of aortic aneurysms and you should stop immediately. Your blood pressure should be checked and if it is persistently raised you should have treatment to reduce your blood pressure. High blood pressure is a risk factor for aneurysm rupture. This does not mean your aortic aneurysm will rupture if you have high blood pressure, but it does place it at slightly higher risk of rupture. It is important to have your risk factors for hardening of the arteries monitored and treated if necessary as there is a clear increased risk of death even after the aneurysm has been treated due to cardiovascular disease (UK Small aneurysm Trial Participants, 2012). There is some evidence that cholesterol lowering drugs such as simvastatin, decrease the growth rate of aneurysms (Schlosser et al 2008).
How will I know if have an aortic aneurysm?
Unfortunately, many people will not know if they have an aortic aneurysm, because they rarely cause symptoms until they burst. Aortic aneurysms are sometimes found during a routine examination for other conditions such as prostate problems or gallstones. If you are a man over the age of 60 years, a smoker with high blood pressure and have a brother or father with an aortic aneurysm, then this puts you at increased risk. If you also have hardening of the arteries at other sites (eg previous stroke or heart attack) then you may also be at increased risk. Occasionally patients present with embolism of blood clot in the aneurysm sac to the lower limb arteries. This occurs when clot is dislodged and travels down to the arteries to the legs and interferes with the blood supply causing scattered areas of mottling in the feet and sometimes gangrene. Although examination by your doctor may be helpful in diagnosing a large aortic aneurysm, it is not a sensitive method of diagnosing smaller aneurysms. The best way to diagnose an aneurysm is with an ultrasound scan of the abdomen. This is a very quick, simple, accurate and safe test that is also commonly used to examine babies in the womb. Occasionally, patients experience abdominal and back pain and the aneurysm becomes tender before it ruptures. If this happens and you know you have an aneurysm, then it is important to seek emergency medical advice. Before surgery most patients will undergo CT (computed tomography) scanning to show the aneurysm in more detail. The picture below is a CT scan of an aneurysm. This picture represents a slice across the abdomen. The spinal column and back is shown in the lower portion of the picture.
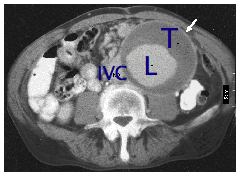
White arrow points to aneurysm
T is thrombus or blood clot inside the enlarged artery
L is the lumen or part of the artery where blood is flowing.
IVC is inferior vena cava or the main vein in the abdomen.
When do aortic aneurysms require treatment?
In healthy people the aorta (the main blood vessel that becomes swollen) is usually about 2.0-2.5 cms (20-25mm) in diameter although this can vary with age and whether you are a man or a woman. We know from two large studies in the USA and UK (Lederle FA et al, 2002) that aneurysms less than 5.5 cms (55mm) across can be safely watched as long as they are monitored on a regular basis. For aneurysms less than 4.4 cms across or less, a yearly ultrasound scan is sufficient to monitor aneurysm growth. For aneurysms between 4.5 and 4.9 cms across, a scan every 6 months is advised. An aneurysm greater than 5.0 cms across requires scans every 3 months although there is some variation in recommendations.
When an aneurysm reaches 5.5 cms most surgeons would consider offering surgical intervention. This is because, at this size, the aneurysm has a greater risk of rupture. It then becomes as safe to have an operation to repair the aneurysm, as it is to leave the aneurysm alone. Surgery may also be considered if your aneurysm is rapidly expanding on regular scans or it starts to cause other complications (see above). Rapid expansion means more than 7mm in 6 months or 10mm in one year.
Whether you proceed with surgery will not just depend on the size of the aneurysm. It is important that each patient is fit enough to withstand the operation. Fitness for surgery can be affected by many factors and the decision whether or not to proceed with surgery can be a difficult one, as it is a very major operation. It will only be after a detailed discussion with your surgeon, regarding your own personal circumstances and type of treatment available, that a decision can be reached.
There is still some debate on the treatment of aneurysms between 4.0 and 5.5cms despite the large UK and North American trials indicating that there is no clear benefit. Looked at in another way though, there was no clear disadvantage to having the aneurysm treated at an earlier stage. Overall 60% of all patients in the trial would eventually require an operation so why not step in at an earlier stage? Taking patients with aneurysms over 5.0cms the argument is even more convincing, as over 80% of these patients eventually require surgery. However, the accepted size to initiate treatment is still 5.5cms (55mm).
Endovascular treatment of aneurysms (see below) has lower operative mortality than open surgery and it was proposed that treating smaller aneurysms with endovascular stents would lead to better outcomes, but in fact the CAESAR and PIVOTAL trials have not shown any advantage for treating smaller AAAs with EVAR. An interesting extra finding was that some of the smaller aneurysms (about 1 in 6) may become unsuitable for EVAR as they grow towards the usual size for intervention, although there is some disagreement on this point. If your aneurysm bursts it usually causes severe back and abdominal pain. The bleeding can stop temporarily in some patients and in these patients an emergency operation can be successful at repairing the aneurysm. The majority of patients (70-80%) with a ruptured abdominal aneurysm will not survive. It is important to remember that although any anxiety you may have had about your aortic aneurysm will be relieved by having it repaired, the operation will not make you feel physically better. This is because most patients do not have symptoms from their aneurysm before the operation. The operation is a treatment to prevent the aneurysm rupturing or causing other complications in the future.
Thoracoabdominal aneurysms
Most (90%) aortic aneurysms are found below the arteries to the kidneys (renal arteries) in the abdomen Some aneurysms can extend upwards to involve the arteries to the kidneys, the arteries to the intestines, liver and stomach. More extensive aneurysms will involve the aorta from where it leaves the heart and can extend throughout the chest. Thoracic (in the chest) aneurysms are probably under-recognised and may involve single or multiple segments of the aorta in the chest. There are rare genetic conditions which can predispose to thoracic aneurysms such as Ehlers-Danlos syndrome and Marfan syndrome. Aneurysms above 6cm carry a significant increased risk of rupture (Kuzmick et al 2012). Thoracic aneurysms are much more difficult to repair and carry much greater risks from treatment. Open surgery for these aneurysms is massive surgery, but until recently was the only treatment available. Newer endovascular treatments are revolutionising therapy in patients with these aneurysms and extending the treatment to patients who previously would have been considered unfit for open repair. Detailed anatomical information is required not only about the aneurysm but also about the aortic branches and their relationship to one another in 3-D. This is a rapidly developing area with different devices and different combinations of treatments being explored.
What treatments are available for aortic aneurysms?
Open Surgery – the traditional treatment for aneurysms is surgery to replace the diseased blood vessel with an artificial blood vessel (graft). In the conventional open operation a large incision is made in the abdomen. The blood vessels above and below the aneurysm are clamped in order to control any bleeding and the aneurysm itself is opened. Any blood clot in the aneurysm is removed and any bleeding blood vessels are controlled. The artificial graft is then stitched into place using permanent stitches (see picture right). The graft is made from a man-made material called Dacron, similar to Terylene.
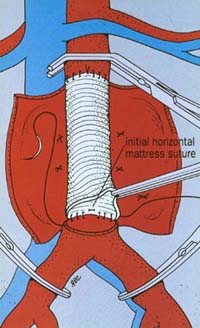
This is a major operation requiring 7-10 days in hospital and usually a short post-operative stay on the intensive care unit. If you have this operation as a planned procedure there is an overall risk of dying of around 5%. This means that 95 patients out of every 100 will be fine and come through the operation. However, a small number of patients (approximately 5 in 100) will die in hospital either during or more commonly after their operation. These are average results when looked at overall on a country wide basis, although some individual units may claim better survival figures. It is important to remember that your chances of surviving a planned operation are much better than if your aneurysm ruptures, when the overall chance of dying is around 80%. This means that 80 patients out of every 100 who have a ruptured aneurysm will die. The conventional open operation has a history dating back over 55 years and is a very effective and durable treatment for aneurysms. Once patients have recovered from the operation most do not have further problems.
Endovascular stenting – over the last 18 years a new treatment has become available (Endovascular Aneurysm Repair or EVAR). This procedure is different from the conventional operation because it does not usually require any cuts in the tummy. Two small vertical cuts are made in the groin and an artificial graft (tailored individually for each aortic aneurysm) is delivered to and deployed from inside the aneurysm itself. This operation requires a special delivery device to deliver the graft through the arteries to the aneurysm. At least 16 different delivery devices have been developed to facilitate this procedure and it is likely that major advances in delivery systems and devices will continue to simplify the operation.
The video opposite shows a simulation of endovascular stent placement. EVAR is a rapidly developing field with robotic enhancements on the horizon. The stenting operation is only suitable for patients with certain shapes (morphology) of aortic aneurysm. The number of patients suitable for EVAR varies to some extent on the expertise of the local unit. Only about 50-60% of aneurysms will be suitable for the endovascular technique, but it has the attraction of being much less traumatic than the open procedure. As modern generation devices are refined, more and more aneurysms are becoming suitable for the endovascular technique. Recovery is faster for most people and it may permit much earlier discharge from hospital. More and more complex graft stents are being developed which will deal not only with abdominal aneurysms but also with complex aortic arch and thoacoabdominal aneurysms involving major branches of the aorta. An endovascular graft can be constructed with branches to supply major arterial vessels (branched graft) or holes (fenestrations) can be created in the side of the graft so a stent can be inserted into the branch (fenestrated graft). Hybrid procedures involve a combination of open and endovascular techniques. These techniques are expanding the population suitable for aneurysm treatment as previously only relatively fit patients could withstand the major surgery involved in repair of complex thoracoabdominal aneurysms.
Laparoscopic aneurysm repair – There are a few centres developing keyhole surgery for aortic aneurysm repair. In principle this operation is the same as open surgery (see above). The aneurysm is approached from the outside and a graft stitched into place. It is different from the open operation because the incisions used in the abdomen are much smaller.
It is not available as an option in most departments and only a few expert laparoscopic surgeons around the world are performing this procedure and it may turn out to be a surgical dead end. It will require considerable development if it is to become routine, but has the attraction of implanting the graft in a traditional way but without a large incision. This would combine the advantages of a minimally invasive and low trauma approach, with the durability and long term freedom from complications of an open repair.
Non-surgical treatments – the ultimate aim of some research is to slow or prevent aneurysm growth, reducing or eliminating the need for surgery. There are no treatments proven to reduce the need for aneurysm surgery but work continues. Beta blockers, such as metoprolol or atenolol, may be helpful. A recent study (Hackam DG et al, 2006) has reported that the use of angiotensin converting enzyme inhibitor drugs (captopril for example) was associated with a reduced risk of aneurysm rupture, but further studies are required.Emergency treatment for ruptured aneurysms – patients who do not die from a ruptured aneurysm in the community may present to hospital as an emergency usually with back and abdominal pain. For most of these patients emergency open surgery is still by far the commonest procedure. There is about a 50-60% chance of surviving the operation but recovery in these circumstances is often prolonged. There are some tertiary centres that are performing endovascular repair for ruptured aneurysms with good results but this requires well manned units with top class radiological/operating facilities and considerable experience of endovascular repair.
What are the results of surgery for aortic aneurysms?
There are two major studies (the Dutch Randomised Endovascular Aneurysm Management (DREAM) trial and the Endovascular Aneurysm Repair Trial-1 (EVAR-1)) that have reported on the endovascular treatment of infrarenal abdominal aortic aneurysms (the commonest aneurysms). These trials have rigorous trial designs and protocols that have demonstrated significant improvements in 30 day mortality in patients undergoing endovascular repair (Prinsen M et al, 2004; Greenhalgh RM et al, 2004) when compared with traditional open repair for abdominal aortic aneurysms.
In the DREAM trial the risk of dying from open surgery was reduced from 4.6% to 1.2% with endovascular stenting at 30 days after operation. EVAR-1 showed a similar reduction in 30 day operative mortality from 4.7% with open repair to 1.7% with endovascular repair. By 1 year after surgery this benefit had disappeared in the DREAM trial patients (Blankensteijn JD et al, 2005). At two years the survival rates were 89.6% in the open surgery patients and 89.7% in the endovascular repair group. The initial advantage of having an endovascular repair only seemed to last for a short period (months) around the time of the operation and was then lost. Follow up data (EVAR Trial Investigators 2010, De Bruin JL et al 2010, EVAR Trial Participants 2005) have confirmed that the initial survival advantage for endovascular repair is not maintained and longer term survival is just as likely with open surgical repair. It may be that open repair precipitates death in some of the more fragile patients who may only have a limited life expectancy because of other serious illnesses. There is also some evidence that kidney function may deteriorate more quickly after EVAR.
EVAR-2 (EVAR Trial Participants, 2010 and 2005) was a UK study which compared best medical treatment for patients with aortic aneurysms against endovascular repair. This study looked at patients who were not fit for open surgery. After 6-8 years of follow up the overall mortality rates were high at 73% (145 of 197) of patients who underwent endovascular repair and 77% for patients treated medically. This was not a statistically different result and the authors conclude that there is no benefit in having aneurysm treatment, even with the less traumatic endovascular technique if patients have other significant illnesses that preclude open repair. The difficulty here is defining what constitutes significant illness and this will definitely vary between different surgeons. For patients at higher risk careful thought on each individual’s risk/benefit from treatment will be helpful.
Despite the benefits clearly demonstrated on early mortality with endovascular repair, there are still two major unresolved issues. They are the durability of the procedure and the problem of endoleak (see below in complications).
The long term durability of endovascular stenting is becoming clearer but there are still unknowns as the technology and indications for intervention change and it is not a risk free alternative to open surgery (Gorham TJ et al, 2004). A review article in the Journal of Vascular Surgery (Rutherford RB and Krupski WC, 2004) concluded that in our present state of knowledge traditional open surgical repair is still to be preferred in younger patients at low operative risk – a view endorsed again in 2005 (Cronenwett JL, 2005). Increasingly this view is being challenged and in most current practice if a patient has an aneurysm suitable for endovascular repair then this treatment will be offered.
Endovascular stenting is here to stay and in fact is the treatment of choice for 50-60% of all patients with aneurysms in the USA (Nowygrod, 2006). Technology will continue to evolve and complication rates are likely to improve further and there is reasonable evidence that they are dropping already.
At present patients who have undergone EVAR require lifelong follow up with imaging of their graft to detect problems at an early stage. This is usually by a combination of CT scan and ultrasound with angiography reserved for treating problems when they occur. Ultrasound is becoming the standard investigation and contrast enhanced ultrasound using second generation contrast agents holds promise but more research is required on accuracy and optimal follow up regimes. One study has suggested that follow up may be targeted as most complications occur in patients who develop symptoms (Karthikesalingam A et al).
Are there complications of treatment?
There are a number of potential problems that can occur after open aortic aneurysm surgery. These fall into 2 main categories: generalised complications and local complications. Both types of complication can prolong the stay in hospital and may be fatal if they are severe enough. There is a risk of dying from open aneurysm surgery of between 3-5%.
Generalised complications – this means problems that can occur away from the site of the operation. They occur because a major operation under general anaesthesia has been performed usually with significant blood loss. The commonest types of generalised complication usually occur in the heart or the lungs. They take the form of angina (chest pain), heart attacks and chest infections. In the majority of patients, especially those having a planned operation, these complications can be successfully treated. The combined presence of heart lung and kidney complications can result in the condition of multi-organ failure (MOF). This is frequently fatal. Another generalised complication that can occur with any operation is Deep Venous Thrombosis (DVT)
Local complications – this means problems related to the site of the operation. The main early problem that can occur is bleeding at the place where the main artery has been joined to the artificial graft. This can be severe. The surgeon will stop all bleeding before completing your operation but sometimes further bleeding can develop during the recovery period – especially within the first 24 hours. If this happens the patient can require a further operation to control the bleeding.
Nerves controlling sexual function run very close to the aorta. Although attempts to preserve these nerves are usually made during an aneurysm repair they can be frequently damaged. In men this can lead to loss of erections. If erections are preserved, retrograde ejaculation can take place where the semen is ejected into the bladder due to incoordination of the various muscles. The effects of damage to the same nerves in women are not clear in the age group that usually require this operation.
Sometime after the operation, infection can develop in the artificial graft, although this is rare. If this does occur it can be a major problem and will probably lead to further surgery to fix the problem. Occasionally replacement of the major artery in the abdomen can lead to impairment of the blood supply to part of the colon (colonic ischaemic). If this becomes very severe then further surgery can be necessary to remove the damaged colon and prevent further complications.
In approximately 30% of patients a weakness can develop in the scar on the tummy. If this happens it occurs months or even years after recovery from the original surgery. It can lead to bulging in the abdominal wound and the development of an incisional hernia. This seems to be more common in aneurysm patients and may require a further operation. Endoleak is a complication virtually exclusive to endovascular aneurysm repair. The behaviour and management of different types of endoleak is becoming more clear cut with increased experience in this area.
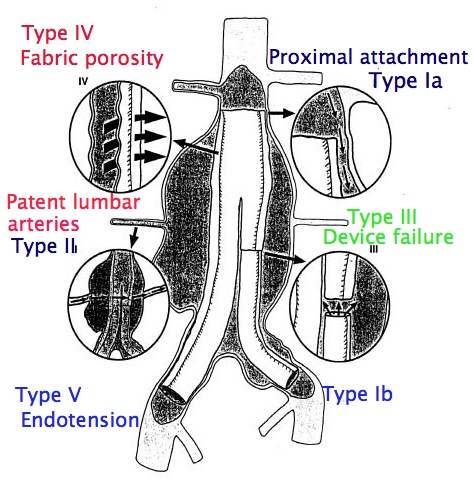
Not all endoleaks demand immediate correction. Endoleak is only a problem in patients undergoing endovascular repair. It occurs when the seal between the graft and the normal arterial wall or between two segments of graft is incomplete (see diagram below (Type I – V endoleaks). If the seal is not complete blood can leak through the seal and fill the aneurysm as it did before the operation. There are also other types of endoleak in which small arterial branches of the aneurysm continue to fill the aneurysm sac. If endoleaks are significant and cause pressurisation of the partly excluded sac (endotension) this can lead to aneurysm rupture – the very thing surgeons are trying to prevent. The EVAR and DREAM trials were initiated many years ago and complication rates may not reflect current practice. There are also varying estimates of the risk of reintervention, but the most reliable results will be from the trials. EVAR-1 showed the risk of complications developing over 6-8 years was 45% (282 of 626) in the endovascular group versus 13% (78 of 626) in the open repair group. Around 20-30% of patients (20-30 in every 100 patients) will require a further procedure over 6-8 years to keep the stent working normally and prevent aneurysm rupture. After open surgery the risk of reintervention over the same time period is about 8-10%. A recent editorial has drawn attention to the 3% ongoing annual failure rate for endovascular grafts. This is in addition to the annual rate of further intervention in conjunction with a need for lifelong follow-up. The author also highlights results from the Cleveland Clinic (a well known US vascular centre) where the results of endovascular grafting in large aneurysms were concerning with a 10% (1 in 10) 4 year risk of rupture for aneurysms 6.5cm in diameter or greater at the time of endovascular repair. In other words, even after apparently successful endovascular repair patients with large aneurysms still had a 1 in 10 chance of their aneurysm rupturing.
What measures are taken to reduce complications?
To reduce the risk of DVT – Anti-embolic graduated compression stockings may be used, providing there is no evidence of hardening of the arteries in the legs. Intermittent compression of the legs and/or feet using airbags is sometimes used in theatre to improve blood flow in the leg veins during the anaesthetic. Most patients also receive heparin injections to reduce the risk of blood clots forming. After your operation you will be encouraged to move around as early as possible.To reduce the risk of infection – Antibiotics will be given at the start of the operation and sometimes for one or two doses after the operation. Physiotherapy will be started shortly after the operation to prevent secretions accumulating in the chest.
Aortic aneurysm screening programmes
As aneurysms can be a serious health issue much effort has been directed at trying to identify aneurysms before they rupture. Screening programmes have been used to detect patients with aortic aneurysms and to monitor the aneurysms if they are small. An aortic aneurysm will usually develop slowly and enlarge over a period of years. Surgery can be considered as the aneurysm enlarges. Although these AAA screening programmes cannot prevent aortic aneurysms forming, they are able to reduce the chances of an aneurysm rupturing by treating it at an early stage.
Aortic aneurysm links
General
http://www.nhs.uk/conditions/repairofabdominalaneurysm/pages/introduction.aspx
http://hcd2.bupa.co.uk/fact_sheets/Mosby_factsheets/aortic_aneurysm.html
http://www.medicinenet.com/abdominal_aortic_aneurysm/article.htm
http://www.emedicine.com/med/topic3443.htm
http://www.surgical-tutor.org.uk/default-home.htm?system/vascular/aaa.htm~right
http://en.wikipedia.org/wiki/Abdominal_aortic_aneurysm
http://en.wikipedia.org/wiki/Aortic_aneurysm
Images
http://www.gvg.org.uk/pics.htm
http://www.vesalius.com/graphics/archive/archtn.asp?VID=664&nrVID=663
http://www.vesalius.com/graphics/archive/archtn.asp?VID=631&nrVID=630
Endovascular Stenting
http://www.uva.vasculardomain.com/images/uploaded/uva/stents.cfm
http://www.dcmsonline.org/jax-medicine/2000journals/dec2000/endovascular.htm
http://www.sirweb.org/patPub/abdominalAorticAneurysms.shtml
http://www.pennhealth.com/int_rad/health_info/aaa.html
http://www.orlive.com/baptisthealth/videos/aortic-aneurysm-repair?view=displayPageNLM
References
Bengtsson H, Bergqvist D, Sternby NH. Increasing prevalence of abdominal aortic aneurysms. A necropsy study. Eur J Surg 1992; 158: 19-23.
Sayers RD. Aortic aneurysms, inflammatory pathways and nitric oxide. Ann Roy Coll Surg Eng 2002; 84: 239-246.
Rossak JI, Sporle A, Birks CL, van Rij AM. Abdominal aortic aneurysms in the New Zealand Maori population. Brit J Surg 2003; 90: 1361-1366.
Filipovic M, Goldacre MJ, Roberts SE et al. Trends in mortality and hospital admission rates for abdominal aortic aneurysm in England and Wales, 1979-1999. Brit J Surg 2005; 92: 968-975.
Sandiford P, Mosquera D, Bramley D. Trends in incidence and mortality from abdominal aortic aneurysm in New Zealand. Brit J Surg 2011; 98: 645-651.
Powell JT. Familial clustering of abdominal aortic aneurysm – smoke signals, but no culprit genes. Brit J Surg 2003; 90: 1173-74.
Weintraub NL Understanding abdominal aortic aneurysms N Engl J Med 2009; 361: 1114-1116.
Brown PM, Zelt DT, Sobolev B. The risk of rupture in untreated aneurysms: the impact of size, gender and expansion rate. J Vasc Surg 2003; 37: 280-284.
UK Small Aneurysm Trial Participants. Final 12-year follow-up of surgery versus surveillance in the UK small aneurysm trial. Brit J Surg; 94: 702-708.
Schlosser FJV, Tangelder MJD, Verhagen HJM et al. Growth predictors and prognosis of small abdominal aortic aneurysms. J Vasc Surg 2008; 47: 1127-33.
Lederle FA et al. Immediate repair compared with surveillance of small abdominal aortic aneurysms. N Engl J Med 2002; 346: 1437-44.
Cao P, De Rango P, Verzini F, Parlani G, et al for the CAESAR Trial Group. Comparison of Surveillance versus aortic endografting for small aneurysm repair (CAESAR): results from a randomised trial. Eur J Vasc Endovasc Surg 2011; 41: 13-25.
Ouriel K, Clair DG, Kemt KC, Zarins CK. Positive impact of endovascular options for treating aneurysms early (PIVOTAL) investigators. Endovascular repair compared with surveillance for patients with small abdominal aortic aneurysms. J Vasc Surg 2010; 51: 1081-7.
Kuzmik GA, Sang AX, ElefteriadesJA. Natural history of thoracic aortic aneurysms. J Vasc Surg 2012;56(2): 565-71.
Gorham TJ, Taylor J, Raptis S. Endovascular treatment of abdominal aortic aneurysm. Brit J Surg 2004; 91: 815-827.
Rutherford RB and Krupski WC. Current status of open versus endovascular stent-graft repair of abdominal aortic aneurysm. J Vasc Surg 2004; 39(5): 1129-1139.
Cronenwett JL. Endovascular aneurysm repair: important mid-term results. Lancet 2005; 365: 2156-58.
Prinssen M, Verhoeven ELG, Buth J et al. A randomised trial comparing conventional and endovascular repair of abdominal aortic aneurysms. New Engl J Med 2004; 351: 1607-18.
Greenhalgh RM, Brown LC, Kwong GP, Powell JT, Thompson SG. Comparison of endovascular aneurysm repair with open repair in patients with abdominal aortic aneurysm (EVAR trial 1), 30 day operative mortality results: randomised controlled trial. Lancet 2004; 364: 843-8.
Blankensteijn JD, de Jong SECA, Prinssen M et al. Two-year outcomes after conventional or endovascular repair of abdominal aortic aneurysms. N Engl J Med 2005; 352: 2398-405.
Evar Trial Participants. Endovascular aneurysm repair versus open repair in patients with abdominal aortic aneurysm (EVAR-1): randomised controlled trial. Lancet 2005; 365: 2179-86.
De Bruin JL Baas AF, Buth J et al. Long term outcome of open or endovascular repair of abdominal aortic aneurysm. N Engl J Med 2010; 362: 1881-9.
The United Kingdom EVAR Trial Investigators.The United Kingdom EVAR Trial Investigators. Endovascular versus Open repair of abdominal aortic aneurysm. N Engl J Med 2010; 362: 1863-71.
The United Kingdom EVAR Trial Investigators. Endovascular repair of aortic aneurysm in patients physically ineligible for open repair. N Engl J Med 2010; 362: 1872-80.
Karthikesalingam A, Holt PJE, Hinchcliffe RJ et al. Risk of reintervention after endovascular aortic aneurysm repair. Brit J Surg 2010; 97: 657-663.
Lederle FA. Abdominal aortic aneurysm – open versus endovascular repair. New Engl J Med 2004; 351: 1677-79.
EVAR Trial Participants. Endovascular aneurysm repair and outcome in patients unfit for open repair of abdominal aortic aneurysm. Lancet 2005; 365: 2187-92.
Nowygrod R, Egorova N, Greco G et al. Trends, complications, and mortality in peripheral vascular surgery. J Vasc Surg 2006; 43: 205-216.
Hackam DG, Thiruchelvam D, Redelmeier D. Angiotensin-converting enzyme inhibitors and aortic rupture: a population-based case-control study. Lancet 2006; 368: 659-65.